Our Science
NRG1 fusions and our “tumor-agnostic” approach
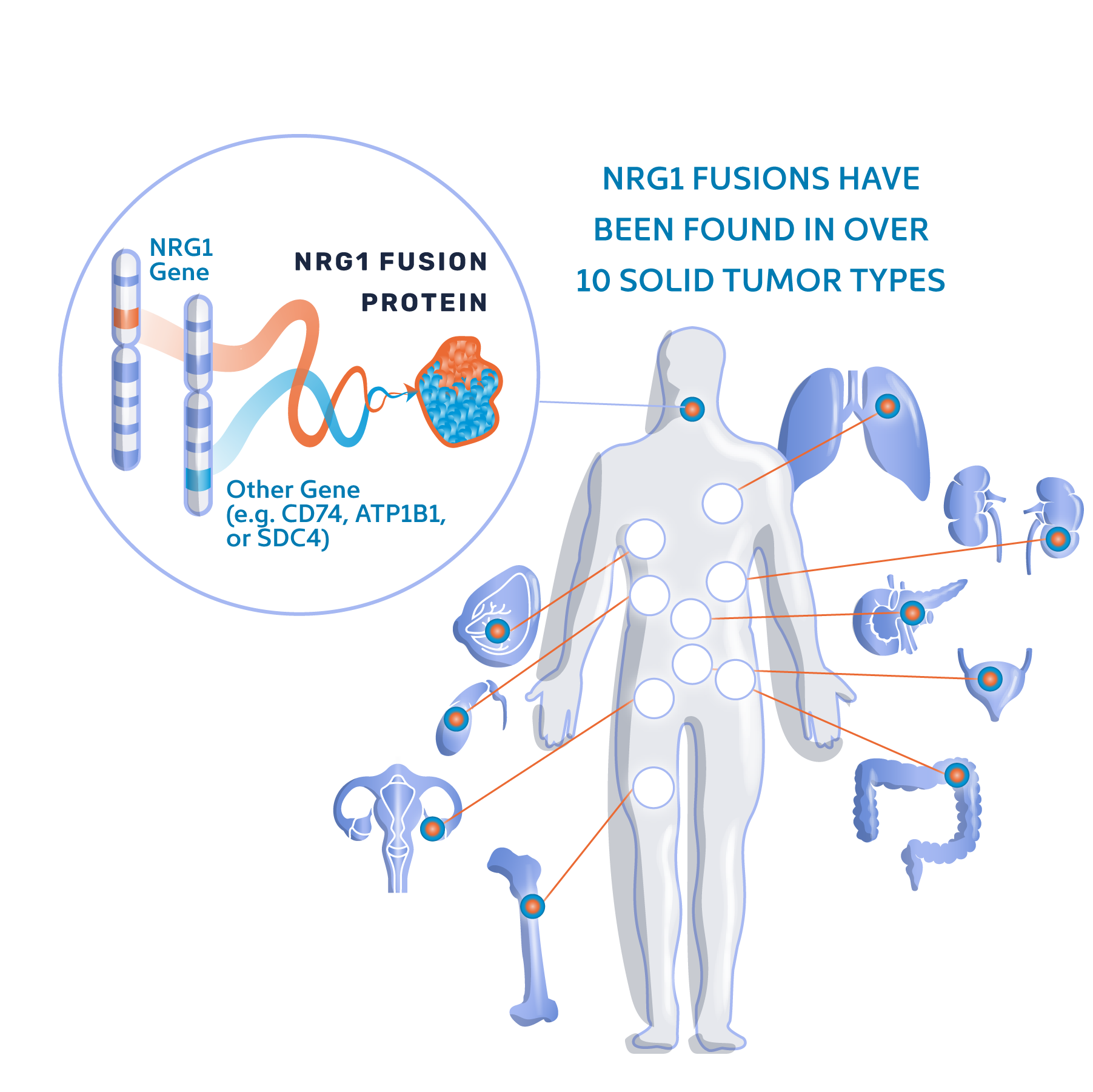
Emerging scientific evidence suggests that NRG1 gene fusions, while rare, are unique driver alterations. When an NRG1 gene fusion is found, inhibiting the HER3 signaling pathway is likely to stop the driving force behind the tumor’s growth and proliferation.
Recently, the FDA has begun to recognize that some precision therapies that target oncogenic driver alterations may be “tumor-agnostic”, meaning they are likely to be effective no matter which organ the tumor is in. Because NRG1 fusions are found in multiple solid tumors and are likely to be a unique oncogenic driver alteration whenever they are found, we have designed CRESTONE as a “tumor-agnostic” study.

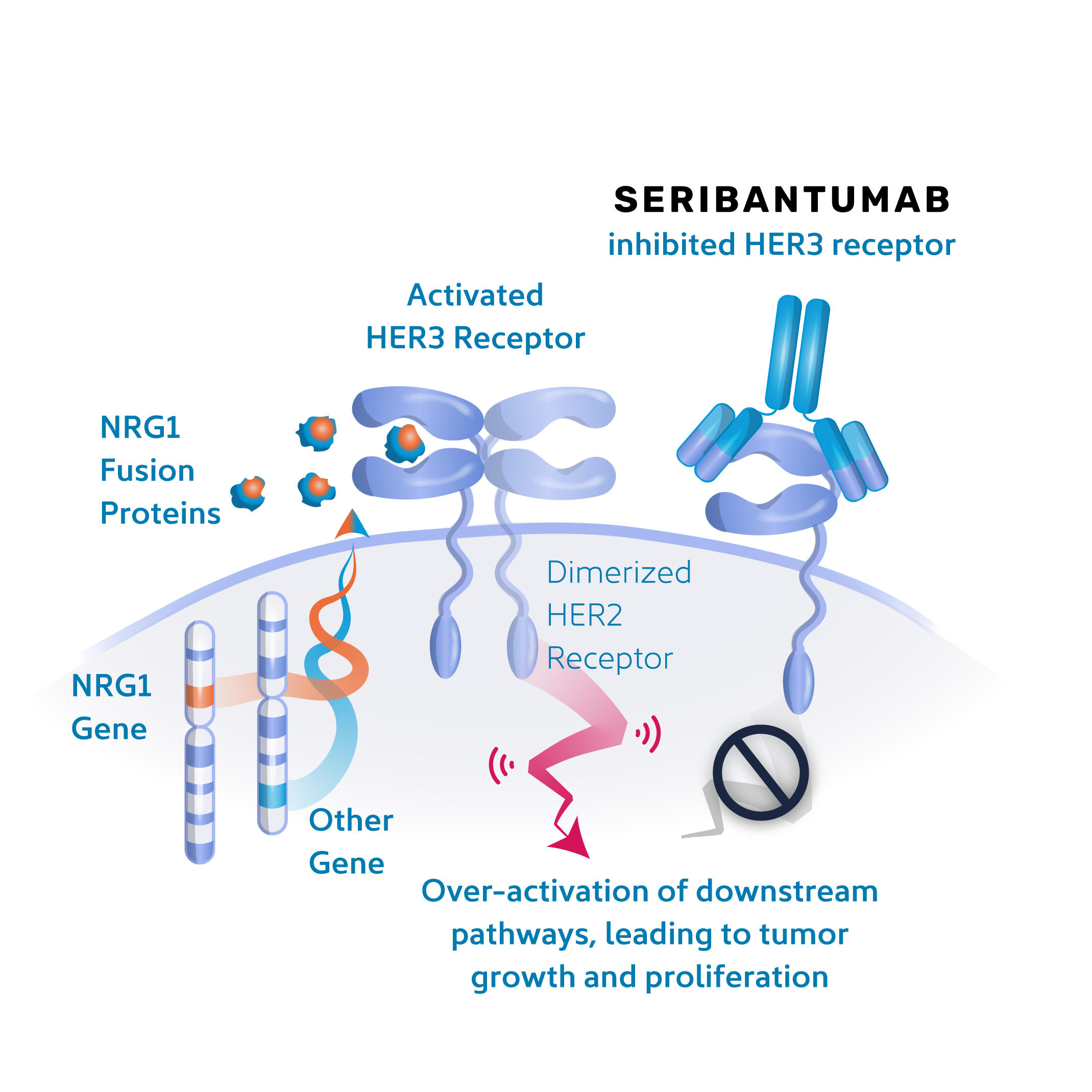
Investigational Treatment: Seribantumab

Seribantumab is an investigational medicine being studied in a Phase 2 clinical trial in patients with solid tumors that have a specific genomic alteration called an NRG1 gene fusion. Seribantumab is believed to work by inhibiting the signaling initiated by the NRG1 fusion protein that tells a tumor cell to grow and proliferate. By stopping the signaling, we may stop the driving force that sustains and promotes the tumor. If you have had your tumor genomically tested and it is NRG1 gene fusion positive, you may be eligible for the CRESTONE study.
Elevation Oncology is the sponsor of the clinical trial and the developer of seribantumab. Detailed information about the study can be found on this site and at www.clinicaltrials.gov (NCT04383210).
“Finding a driver mutation is unlikely, but if one is found, it makes a big difference in management.”
DR. ROBERT DOEBELE, MD PHD, Targeted Oncology, January 2020
Talk with your physician about genomic testing and clinical trials
If you are interested in getting your tumor tested and learning about investigational therapies that may be uniquely targeted to your tumor, please talk to your physician. We encourage all patients to consider genomic testing for their tumor, as this can reveal information that is critical to help guide your treatment plan.
Your physician can help determine if participating in a clinical trial may be an option for the treatment of your cancer. Clinical trials are designed for the investigation of experimental medicines such as seribantumab. As an experimental medicine, seribantumab has not yet been approved for sale by any regulatory authority anywhere in the world, and has not yet been determined to be safe and effective for the purpose for which it is under investigation. There are no currently approved therapies for the specific treatment of patients with an NRG1 gene fusion.
Your decision to participate in a clinical trial is voluntary and should only be made after all your questions have been answered and you have been able to make a well-informed decision.
We are also here to help and provide the information necessary to make an informed decision. You can always contact our medical staff at medicalaffairs@elevationoncology.com or +1 (716) 371-1125.
